
Scientists at SESAME have developed a safer, faster, and more energy-efficient way to trap and solidify toxic chromium from polluted water.
Industrial waste commonly contains chromium, a heavy metal that poses a serious environmental threat. But a study led by a team of researchers at SESAME offers hope, showing that calcium hydroxyapatite (HA) powder can be used to lock chromium safely away, preventing it from leaching into the environment.
Dr. Sajid Iqbal, the lead scientist of the study, expressed that this method has the potential to revolutionize waste treatment. “By using HA, we can capture and safely immobilize chromium, making it far less harmful to the environment and human health.”
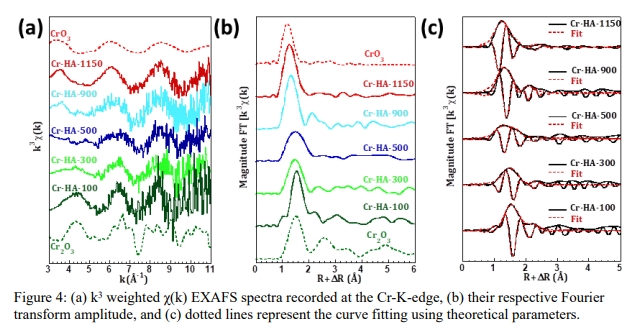
The team’s research focused on understanding how chromium behaves at high temperatures and how it interacts with the HA matrix. They used synchrotron X-ray absorption fine structure (XAFS) spectroscopy to find that sintering (heating the material to very high temperatures) could change the form of chromium inside the HA. Chromium underwent a transformation from a safer, less toxic form (Cr(III)) to a more dangerous one (Cr(VI)) when temperatures increased from 100°C to 1150°C.
“The results were striking,” Dr. Iqbal explained. “At lower temperatures, chromium remains in its less harmful form, Cr(III). But as the temperature rises, it transforms into the more toxic Cr(VI).”
This transformation could pose a problem if the temperatures are too high during the solidification process, as it could release more toxic chromium into the environment. The researchers did find a simple solution, though: they were able to slow down the change from Cr(III) to Cr(VI) by using low-temperature sintering methods and adding certain binders to the HA composite.
“The key takeaway is that with careful control over the sintering temperature and the right additives, we can prevent chromium from becoming more dangerous,” Dr. Messaoud Harfouche. “This makes the process greener and safer for both the environment and people.”
This new approach could revolutionize the way industries manage chromium waste. Instead of releasing toxic chromium into landfills or rivers, companies could use this method to trap it safely, reducing pollution and preventing harmful environmental impacts. The team’s work could be especially important in areas with high levels of industrial waste, where chromium contamination is a major concern.
“This method is not just safer; it’s also more energy-efficient,” Dr. Harfouchesaid. “By reducing the need for high temperatures, we’re saving energy and cutting down on greenhouse gas emissions.”
The team hopes their findings will encourage further research and improvements in the field of waste treatment. Their work is an important step forward in addressing one of the most persistent environmental issues today.
“As we move forward, we plan to refine the technique and explore other ways to make the process even more effective and sustainable,” Dr. Iqbal explained. “We believe this is just the beginning of a greener, safer approach to industrial waste management.”
With pollution and environmental sustainability issues rising, this research offers a possible solution to chromium contamination. It reminds us that simple waste management modifications can drastically improve environmental protection.
